 This is the most common case you will encounter in chemistry.ģ) At very high energies we can get the atoms of different elements moving so fast so that when they collide their nuclei can fuse into one larger nucleus. With a chemical reaction even though chemical bonds have been broken and reformed to create a new chemical, we haven't touched the protons in the nucleus and therefore haven't changed the elements involved. Which as we know is a chemical compound composed of the elements hydrogen and oxygen. Using the hydrogen and oxygen gas example, if we added those gases together and introduced a source of heat like a flame, they would react and combine to form water. Now there is enough energy to break chemical bonds and conduct a chemical reaction where the atoms of different elements can combine into a compound composed to atoms from both elements. For example, if I combine hydrogen and oxygen gas together at low temperatures then I just have a vessel filled with unreacted gas.Ģ) At higher energies we start creating the situation suitable for reactions to occur. You just get a mixture of different types of atoms. Combining two atoms of different elements together depends on how much energy you put into such a collision.ġ) At low energies (think low temperatures essentially) putting together two samples composed of atoms of different elements doesn't usually create a reaction. So in practice we're not really combining elements rather atoms of different elements. This requires less class time to complete thus providing more valuable class time to learn other topics.So elements are different types of atoms which are differentiated by their atomic number (the number of protons). The “Color Coding the Periodic Table” WS has mini Periodic Tables.Show videos/play songs about the Periodic Table while students color.Students can research on the internet how to color the PT.The key can be projected on a screen for students to see as they color code their own Periodic Table. 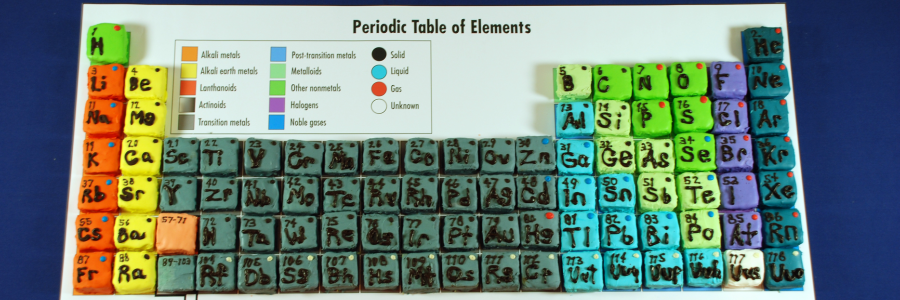
Students can then use the key as a guide for color coding their own Periodic Table. The key can be color printed and laminated to make a class set.Repeat the process again for coding solids, liquids, and gases at room temperature. Remind students to fill in the key with the colors they chose for each. Document cannot be edited.)ĭirections: Instruct students to color code metals, nonmetals, and metalloids on the periodic table. Ĝolor Coding Solids, Liquids, & Gases on Periodic Table WS – KEY.Ĝolor Coding Solids, Liquids, & Gases on Periodic Table WS.Ĝolor Coding Metals, Nonmetals, & Metalloids on Periodic Table WS – KEY.Ĝolor Coding Metals, Nonmetals, & Metalloids on Periodic Table WS.Ĝolor Coding the Periodic Table WS – KEY.Ĝolor Coding the Periodic Table WS (contains a mini Periodic Table for coding solids, liquids, and gases & contains a mini Periodic Table for coding metals, nonmetals, and metalloids).Items: (Items include mini periodic tables and large periodic tables for color coding and keys.) Ĝolor code states of matter (solid, liquid, gas) at room temperature on the Periodic Table.Ĝolor code the metals, nonmetals, and metalloids on the Periodic Table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
